 
Iodine is more likely to gain one electron. Selenium is more likely to gain two electrons. Answer 6Īn Al atom is more likely to lose three electrons. Answer 5Ĭa atom is more likely to lose two electrons. The electron configuration of a oxygen atom is. While core electrons are not involved in bonding, they influence the chemical reactivity of an atom. Positive charges repel each other, so an ionic compound is not likely between two positively charged ions. Generally, valence electrons can participate in the formation of chemical bonding, but core electrons cannot. Ionic bonds are formed by the attraction between oppositely charged ions. The octet rule is the concept that atoms tend to have eight electrons in their valence electron shell. Do you think it will lose seven electrons or gain one electron to obtain an octet in its outermost electron shell? Write the formula of the resulting ion. An iodine atom has seven valence electrons. Do you think it will lose six electrons or gain two electrons to obtain an octet in its outermost electron shell? Write the formula of the resulting ion.Ĩ. A selenium atom has six valence electrons. Do you think it will lose three electrons or gain five electrons to obtain an octet in its outermost electron shell? Write the formula of the resulting ion.ħ. An aluminum atom has three valence electrons. Do you think it will lose two electrons or gain six electrons to obtain an octet in its outermost electron shell? Write the formula of the resulting ion.Ħ. One can use the periodic table to predict whether an. (b) A sodium cation (Na+) has lost an electron, so it has one more proton (11) than electrons (10), giving it an overall positive charge, signified by a superscripted plus sign. A calcium atom has two valence electrons. 1: (a) A sodium atom (Na) has equal numbers of protons and electrons (11) and is uncharged. Why is an ionic compound unlikely to consist of two negatively charged ions?ĥ. Why is an ionic compound unlikely to consist of two positively charged ions?Ĥ. 
In the case of period 2, there are two core electrons in the #"1s"# subshell.įor the transition metals, determining valence electrons is tricky because they can use inner electrons as valence electrons.\)ģ. The rest of the electrons are the core electrons. 10":#"1s"^2"2s"^2"2p"^6"#įor all of the preceding elements, the valence electrons are the outermost (highest energy) #"s"# and #"p"# electrons. A neutral atom has the same number of electrons as protons. The atomic number is the number of protons in the nuclei of the atoms of an element. You can also find the core and valence electrons by determining or looking up the electron configurations of the main group elements. 
Across a period, elements in group 1/IA have one valence electron, elements in group 2/IIA have two valence electrons, elements in group 13/IIIA have three valence electrons, and so on, ending with group 18/VIIIA, which have eight valence electrons, which is the maximum number of valence electrons. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to cesium and francium which are the least electronegative at 0.7. The Pauling scale is the most commonly used. You can determine the number of valence electrons in the atoms of the main group elements by the group number of the element. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The core electrons are in the inner shells and do not participate in chemical reactions. 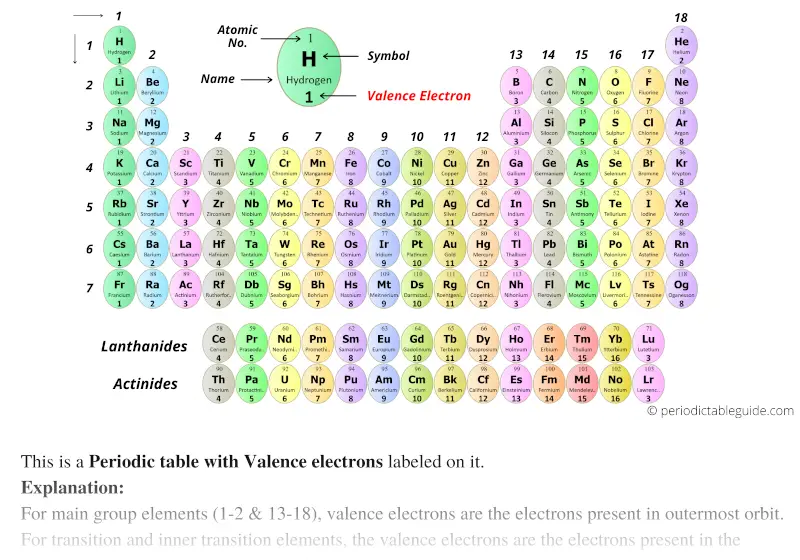
The main group elements are the A groups, or groups 1,2,13-18. The valence electrons participate in chemical reactions. For the main group (representative) elements, the valence electrons are the outermost (highest energy) #"s"# and #"p"# electrons, which make up the valence shell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
